-

-
 SOLVED: the critical of micronutrients needed in tissue equal to or less than 1 ppm (2) 10 (3) 100 ppm (4) 1000 ppm
SOLVED: the critical of micronutrients needed in tissue equal to or less than 1 ppm (2) 10 (3) 100 ppm (4) 1000 ppm
-
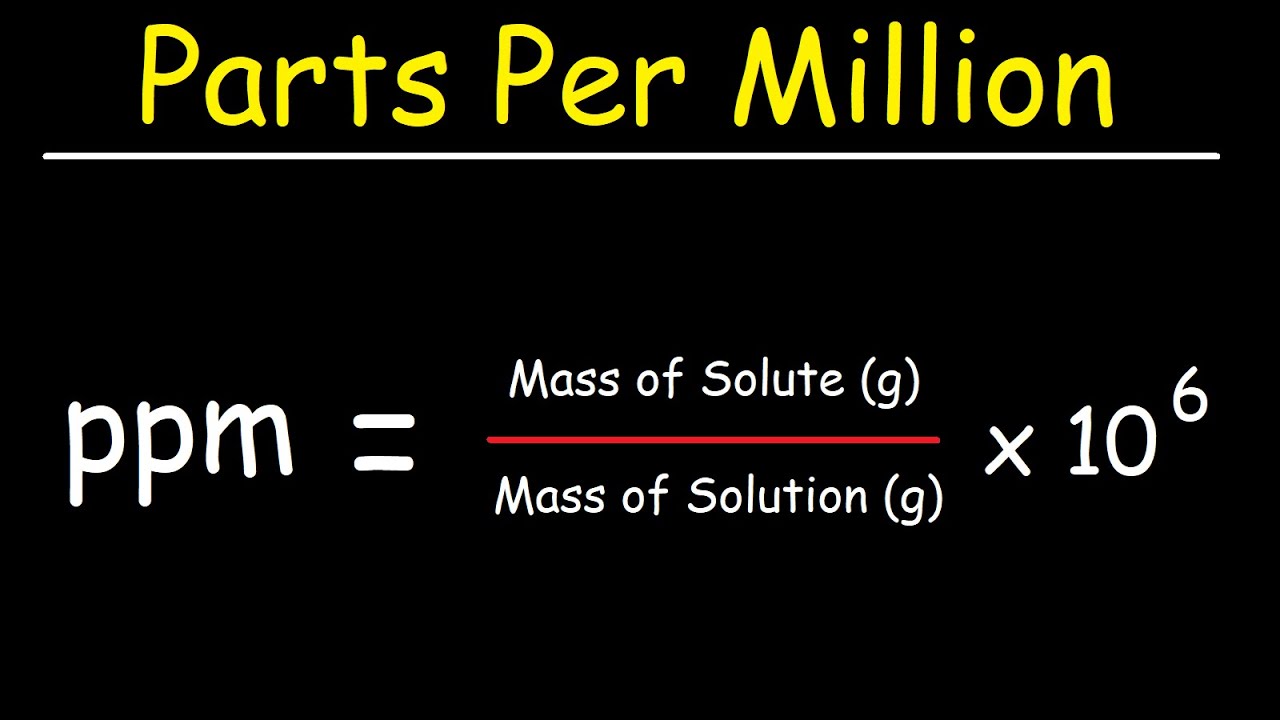 Parts Per Million (ppm) and Parts Per - Solution Concentration - YouTube
Parts Per Million (ppm) and Parts Per - Solution Concentration - YouTube
-
Parts Per (PPM) 3 minutes.
-
 what is ppm - Chemistry Some Basic Concepts of Chemistry - 3782 |
what is ppm - Chemistry Some Basic Concepts of Chemistry - 3782 |
-
 OneClass: A solute is present in aqueous solution at a concentration of 1 ppm mass. What is this
OneClass: A solute is present in aqueous solution at a concentration of 1 ppm mass. What is this
-
 Parts per Million (PPM) Chemical Dilution Formulas General Cleaning
Parts per Million (PPM) Chemical Dilution Formulas General Cleaning
-
 SOLVED: clinical applications, unit parts per million (ppm) is used to express very small concentrations of solute, ppm equivalent to mg of solute per 1 L of solution. Calculate
SOLVED: clinical applications, unit parts per million (ppm) is used to express very small concentrations of solute, ppm equivalent to mg of solute per 1 L of solution. Calculate
-
 SOLVED: In clinical applications, the unit parts per million (ppm) is to express very small concentrations of solute, where 1 ppm1 ppm is equivalent to mg1 mg of solute per
SOLVED: In clinical applications, the unit parts per million (ppm) is to express very small concentrations of solute, where 1 ppm1 ppm is equivalent to mg1 mg of solute per
-
 to Calculator
to Calculator
-
 Concentration of solutions Parts per million (ppm) is a way of expressing dilute concentrations of substances. Just as cent means out of. - ppt download
Concentration of solutions Parts per million (ppm) is a way of expressing dilute concentrations of substances. Just as cent means out of. - ppt download
-
Can be to mg/L?
-
PPM to Calculator - everything RF
-
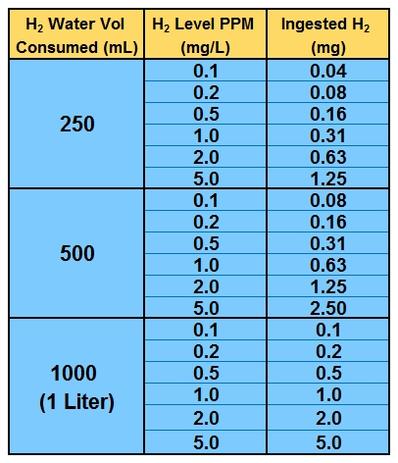 Part Million
Part Million
-
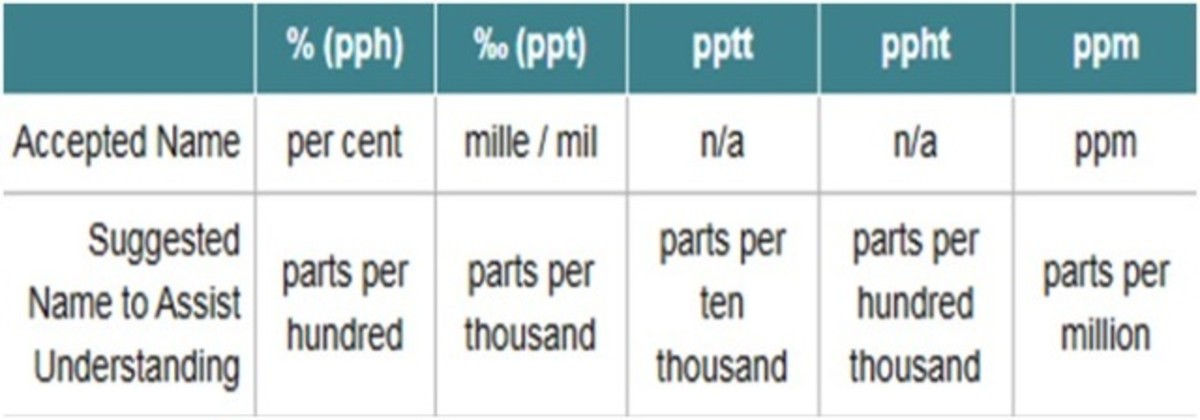 What Does Mean? -
What Does Mean? -
-
 Parts-per notation -
Parts-per notation -
-
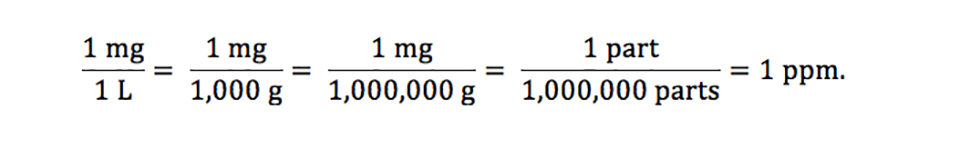 Understanding variables for - Responsible Seafood Advocate
Understanding variables for - Responsible Seafood Advocate
-
 do you convert to - Chilean Way
do you convert to - Chilean Way
-
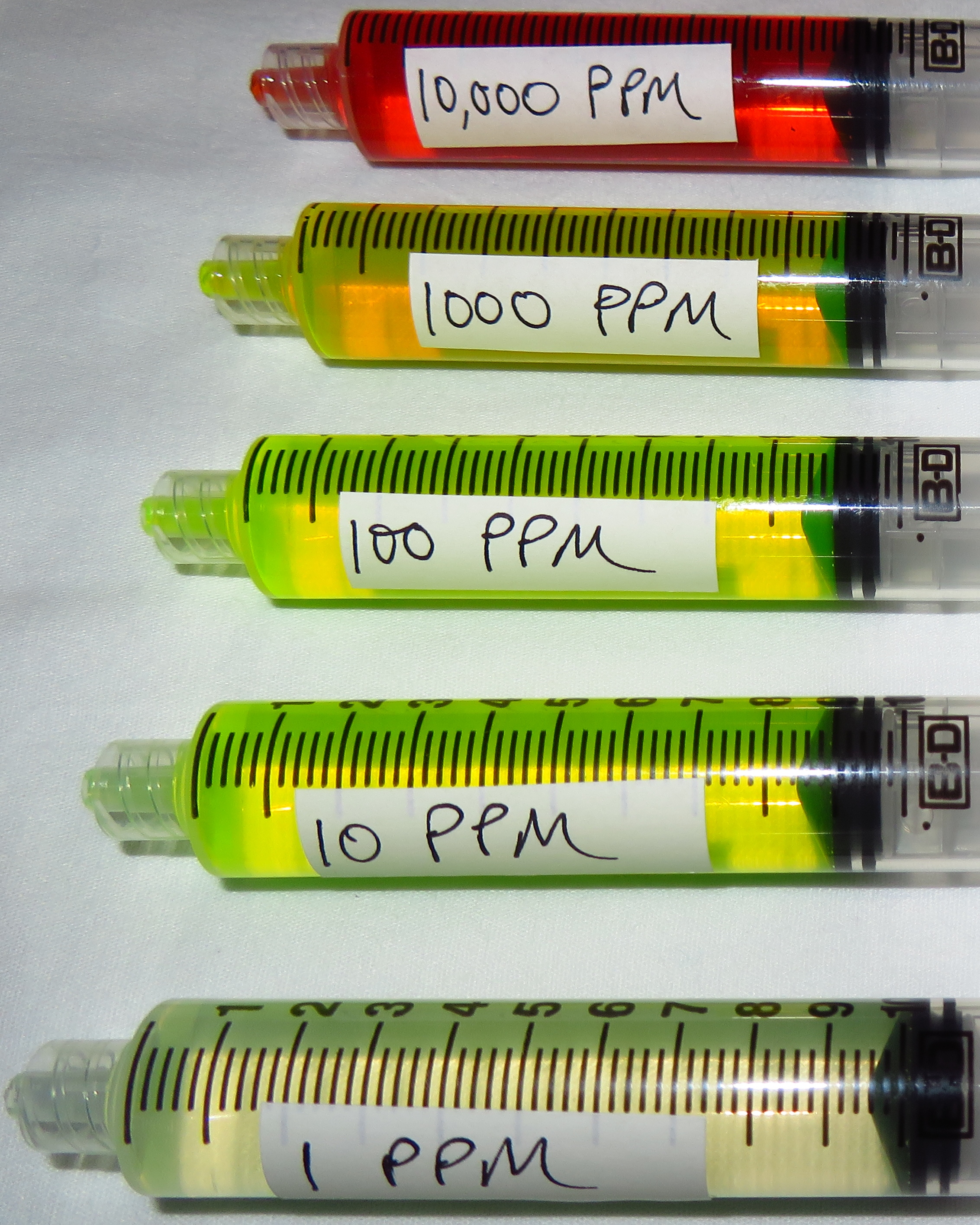 Parts-per notation -
Parts-per notation -
-
 Parts per - ppm
Parts per - ppm
-
 Units There are 1,000 milligrams (mg) in 1 gram (g) milli = 1 milligrams per liter (mg/l) equal to parts per million (ppm) - ppt download
Units There are 1,000 milligrams (mg) in 1 gram (g) milli = 1 milligrams per liter (mg/l) equal to parts per million (ppm) - ppt download
-
 How To Interpret Water Testing – SimpleLab
How To Interpret Water Testing – SimpleLab
-
 ( Everything possible )
( Everything possible )
-
 one ppm to------
one ppm to------